Persistence of the “Moving Things Are Alive” Heuristic into Adulthood: Evidence from EEG
Abstract
Although a growing number of studies indicate that simple strategies, intuitions, or cognitive shortcuts called heuristics can persistently interfere with scientific reasoning in physics and chemistry, the persistence of heuristics related to learning biology is less known. In this study, we investigate the persistence of the “moving things are alive” heuristic into adulthood with 28 undergraduate students who were asked to select between two images, one of which one represented a living thing, while their electroencephalographic signals were recorded. Results show that N2 and LPP event-related potential components, often associated with tasks requiring inhibitory control, are higher in counterintuitive trials (i.e., in trials including moving things not alive or nonmoving things alive) compared with intuitive ones. To our knowledge, these findings represent the first neurocognitive evidence that the “moving things are alive” heuristic persists into adulthood and that overcoming this heuristic might require inhibitory control. Potential implications for life science education are discussed.
INTRODUCTION
Since the late 1970s, it has been thoroughly discussed that students hold many conceptions about natural phenomena that are not consistent with scientific knowledge (Nussbaum and Novick, 1982; Vosniadou, 1994; Carey, 2000; Murphy and Alexander, 2008; Potvin, 2013), these are often referred to as “naive conceptions” or “misconceptions.” In 2007, Duit reported nearly 7700 studies documenting the existence of such nonscientific conceptions in science. For educators and education researchers, these particular ideas are important to consider, as they can interfere with the learning of scientific concepts and can therefore make formal instruction less efficient (Carey, 2000; Liu, 2001). Among the most emblematic examples of such common conceptions is the belief that “bigger or heavier objects sink more,” which can interfere with solving problems about buoyancy (Wandersee et al., 1994; Murphy and Alexander, 2008; Unal, 2008); or that “warmer seasons are caused by an increased proximity between the Earth and the Sun” (Küçüközer, 2008). Another common example can be observed among students when learning about electricity: they often tend to think that lighting can be generated using only a single wire between a battery and a bulb, thus neglecting the concept of circuit (Periago and Bohigas, 2005; Çepni and Keles, 2006; Masson et al., 2014). Interference caused by misconceptions can also affect other areas in science, technology, engineering, and mathematics (STEM), such as mathematics, for instance, when the salient area of a figure interferes with correct evaluation of the perimeter (Stavy and Babai, 2008, 2010; Babai et al., 2016). Furthermore, it has been shown that misconceptions also persist after being addressed by formal instruction (Shtulman and Valcarcel, 2012), making it hard to foster conceptual change (diSessa, 2006).
Several models have been proposed to describe the process of conceptual change (for a review, see Potvin et al., 2020), which can be defined as “learning pathways from students’ pre-instructional conceptions to the science concepts to be learned” (Duit and Treagust, 2003, p. 673). While a number of early models suggested that learning science concepts requires replacing initial intuitive conceptions with scientifically accurate ones (Nussbaum and Novick, 1982; Potvin 2013), others suggest that cognitive resources, such as intuitive rules (Stavy and Tirosh, 2000), core intuitions (Brown, 1993), or phenomenological primitives (diSessa, 2006), must be integrated and organized into a knowledge system during conceptual change in order to acquire the competency to use the appropriate resources correctly in each situation. According to these resource models, pieces of knowledge related to misconceptions are not necessarily erased, eradicated, or replaced during conceptual change but are instead integrated into a wider and more complex system.
More recent contributions have focused on the fact that misconceptions can coexist with scientifically correct knowledge in an individual mind (Dunbar et al., 2007; Bélanger, 2008; Ohlsson, 2009; Shtulman and Valcarcel, 2012) and that cognitive resources can be incorrectly used, even if they have been integrated into a valid knowledge system. According to this perspective, some particular conceptions would persist despite successful learning of scientific conceptions, because they rely on deeply anchored heuristics that have not been erased or replaced during conceptual change (De Neys and Goel, 2011; Shtulman and Valcarcel, 2012; Potvin and Cyr, 2017), perhaps because they are frequently reinforced in everyday life (Ohlsson, 2009). Heuristics are typically defined as “strategies that are effortless, rapid, often global or holistic which constitute the most adaptive response in most situations but sometimes they are misleading especially in situations in which they compete with logical algorithms” (Houdé and Borst, 2015, p. 2). Heuristics are thus particularly useful, because they allow fast and spontaneous reasoning and decision making (i.e., “fast thinking” in Daniel Kahneman’s words; Kahneman, 2011). Children and adults tend to use their heuristics spontaneously and unconsciously (Houdé, 2000). Indeed, although several studies suggest that the brain is able to detect that a heuristic is possibly misleading, its use remains very automatic and requires an additional, but sometimes imperceptible effort to be suppressed (for a discussion, see De Neys, 2014). In the context of science education, we will hereafter identify as intuitive conceptions those nonscientific conceptions that can be based on the automatized use of “fast-and-frugal” (Gigerenzer and Todd 1999) heuristics. For example, because we can feel that a heavier object pushes harder on the hand that holds it, this observation may help explain the widespread use of the “bigger objects sink more” heuristic (Unal, 2008), which could contribute to an intuitive conception of buoyancy.
Because of the usefulness and strength of such heuristics, Potvin (2013) further argues that, from a pedagogical point of view, the coexistence of both scientific and intuitive conceptions implies that developing expertise/learning should be more about making appropriate conceptions prevail, rather than investing teaching time in trying to make students reject and replace useful—albeit not always accurate—conceptions. This coexistence perspective suggests that intuitive and scientific conceptions may in fact compete with one another during the learning process and possibly even throughout life (Dawson, 2014), an idea that is in line with dual-process models that characterize human reasoning as relying on two types of thinking: system 1 (intuitive, effortless, and fast) and system 2 (slower, effortful, but sometimes more accurate; Evans, 2010; Evans and Stanovich, 2013; Kahneman, 2011).
Therefore, educators and researchers alike are looking for means to help students select the appropriate scientific conceptions over the associated intuitive ones (Masson et al., 2014; Wilkinson et al., 2019; Zhu et al., 2019). In this sense, literature suggests that inhibitory control would play a role in scientific reasoning by allowing the individual to suppress a tempting but inaccurate answer in favor of a less intuitive but more scientific one when the context requires it (Stavy and Babai, 2010; Babai et al., 2012; Brault Foisy et al., 2015, 2021; Nenciovici et al., 2019). Inhibitory control represents the ability to resist automatisms, temptations, distractions, or interference and to adapt to conflicting situations (Diamond, 2013). It is thus considered to be a core process of cognitive and socioemotional development (for a review, see Borst et al., 2015). The mechanism of inhibition comes into play when it is necessary to overcome responses that are habitual or overlearned in order to select an alternative response (van den Wildenberg and van der Molen, 2004). As is the case for several other fields of learning (Diamond, 2013; Borst et al., 2015), such as fractions (Meert et al., 2010), arithmetic (Espy et al., 2004; Gilmore et al., 2013), grammar (Wan et al., 2008), and reading (Borst et al., 2015), inhibitory control would also represent a central mechanism in scientific reasoning for overcoming intuitive conceptions that are based on deep heuristics (Allaire-Duquette et al., 2019, 2021; Zhu et al., 2019). In this work, we refer to inhibitory control as a process that allows one to reason logically and thus scientifically by suppressing misleading heuristics from system 1 when they interfere with the activation of the logical algorithms from system 2 (i.e., in our context, the scientific conception; Houdé and Borst, 2015).
Some heuristics can be quite persistent, even after studying science for many years. For instance, it has been argued that both undergraduate students and professional scientists persistently tend to endorse naive explanations when having to answer a question in a short amount of time (Kelemen and Rosset, 2009; Kelemen et al., 2013). In fact, even for very basic conceptions mastered at an early age, such as buoyancy of objects, adults still tend to take more time to correctly evaluate a counterintuitive but scientifically accurate claim (Babai and Amsterdamer, 2008). In this context, longer response times have been interpreted as an indication of the complexity of the reasoning involved: a longer response time could therefore indicate that evaluating correctly counterintuitive claims requires greater cognitive effort to inhibit intuitive conceptions (Kelemen et al., 2013; Potvin et al., 2014). Such behavioral results have often been complemented by studies using neuroimaging techniques, such as functional magnetic resonance imaging (fMRI), showing that selecting a scientific conception instead of using a very tempting but inaccurate heuristic is associated with greater activation in regions of the brain known to be involved in inhibitory control, such as the anterior cingulate cortex or the ventrolateral prefrontal cortex (Masson et al., 2014; Brault Foisy et al., 2015; Allaire-Duquette et al., 2021). Inhibition occurs in the brain through inhibitory neurotransmitters that are released by neurons of the prefrontal region and that can temporarily stop a neuron or a group of neurons from activating and producing an action potential (Ward, 2015). The vast majority of the studies that have established an association between inhibitory control and scientific reasoning, using both response times and psychophysiological methodologies such as fMRI (Brault Foisy et al., 2015; Masson et al., 2014) and electroencephalography (EEG; Zhu et al., 2019), targeted physics as their domain of interest. To a lesser degree, conceptions from other scientific fields such as chemistry have also been studied with the idea of inhibition in mind (Babai and Amsterdamer, 2008; Malenfant-Robichaud, 2018). However, to date, much less research has been carried out regarding the possible role of inhibition in learning life sciences, which is rather surprising, given the fact that they are a central part of most school curricula.
In biology, previous work has highlighted that some conceptions about living things can often be affected by the “moving things are alive” heuristic, which consists in assessing (often unconsciously) whether a thing is alive or not based on its eventual mobility (Opfer and Siegler, 2004). For example, classifying a plant in the living category has been proven to be more difficult than classifying an animal in the same category, because a plant does not move, unlike animals (Stavy and Wax, 1989; Babai et al., 2010). It has been suggested that this heuristic would result from generalizations of human features by young children to imply life, such as psychological capacities or other human features, including mobility (Carey, 1985; Slaughter et al., 1999). However, results indicate that this heuristic is present not only in young children but also in adolescents. Indeed, while it appears that teenagers can easily and accurately identify what is alive and what is not, response-time studies show that correctly identifying a living but nonmoving thing as alive takes more time compared with correctly identifying a living and moving thing as alive (Babai et al., 2010). This frequent “moving things are alive” heuristic is particularly interesting from a pedagogical point of view, as one would tend to expect it to vanish after a certain age because of its obvious falseness. It therefore seems relevant to seek a better understanding of the persistence of this heuristic and the possible role of inhibitory control in overcoming it at different ages.
While the link between inhibitory control and the “moving things are alive” heuristic has already been studied using response-time methodologies with adolescents (Babai et al., 2010; Brault Foisy et al., unpublished data), the results have not yet been corroborated using psychophysiological measurements in adult participants. Using EEG in addition to response times allows triangulation of data and interpretation. Such triangulation is nevertheless essential, because inhibitory control represents a complex cognitive process that can only be measured through inference from indirect measures. The interpretation of response-time studies is limited by several confounding factors, for instance, age (Williams et al., 2005) or task constraints and characteristics (Verguts et al., 2011; Potvin et al., 2014).
This paper aims to evaluate the persistence of the “moving things are alive” heuristic into adulthood using response times and psychophysiological measurements. Specifically, EEG was selected for this study, as it allows for the detection of brain signal variations that can be related to inhibitory control (e.g., Bokura et al., 2001; Harper et al., 2014). Several studies have indeed identified EEG patterns as an index of inhibitory control: the N2 (or N200) event-related potential (ERP) component (observed at around 200 milliseconds) and the late positive potential (LPP) component (see, e.g., Huster et al., 2010; Zhu et al., 2019). More precisely, an N2 component is considered to reflect cognitive processes such as error detection and conflict monitoring (Folstein and Van Petten, 2008; Dickter and Bartholow, 2010; Bockler et al., 2011; Spapé et al., 2011; Liu et al., 2015). Its amplitude is usually larger for intuitive stimuli involving an error or conflict (Zhu et al., 2019). The latency of the N2 component is not clearly agreed upon in the literature. While some authors go so far as to position it between 140 and 200 milliseconds (Colasante et al., 2017), especially for visual stimuli (Folstein and Van Petten, 2008), others place it at a later latency of 250–320 milliseconds (Daurignac et al., 2006), with a spectrum between those extremes (Bahramali, 1999; Patel and Azzam, 2005; Schmajuk et al., 2006; Enriquez-Geppert et al., 2010; Pasion et al., 2019).
While associated with a broad number of cognitive processes, the LPP component is used as a measure of cognitive processes such as error recognition, conflict resolution, and response selection (Mecklinger, 2000; Paller et al., 2007; Voss and Paller, 2009; Coderre et al., 2011; Yin et al., 2016). Moreover, these components have been associated with tasks requiring inhibitory control (e.g., Bokura et al., 2001; Harper et al., 2014). The latency of the LPP is also not a matter of consensus in the literature. To our knowledge, LPP can be found in latencies ranging from 300–450 milliseconds (Enriquez-Geppert et al., 2010) to 500–800 milliseconds (Colasante et al., 2017), with other latencies ranges between those extremes (Wood and Kisley, 2006; Bertoli and Bodmer, 2016; Pasion et al., 2019).
Considering previous evidence, these ERP components can serve as potential indices of the persistence of a given heuristic and the need to inhibit it to perform a task correctly.
Based on these previous studies, we targeted the following research question: Can we observe a difference in terms of behavioral results and EEG patterns between intuitive and counterintuitive stimuli that could be an indicator of inhibitory control and persistence of the “moving things are alive” heuristic in adult participants? We formulate the following hypotheses:
Longer reaction times and lower accuracy rates will be observed when identifying things that are alive but not moving as living things (counterintuitive condition [CI]), compared with when identifying things that are alive and moving (intuitive condition [I]).
Significant differences at the N2 and LPP components will be observed for the same comparison (CI > I).
MATERIAL AND METHODS
Participants
Twenty-eight undergraduate students (13 females, 15 males) aged between 18 and 35 years old (M = 23.7, SD = 0.3 years) were recruited to take part in this study. All of them were registered in a French-speaking business school. All participants were right-handed and had normal vision or appropriate lenses to allow them to work properly on a computer. No participant reported any neurological condition. EEG data collection took place at Tech3Lab (HEC, Montreal). The recruitment was carried out via an online platform of the Tech3Lab, which described the goals of the study, the procedure, the inclusion criteria, and the monetary compensation for participation.
Experimental Procedure
Upon arrival, each participant received a brief verbal description of the research project, as well as the general procedure to come, and was then asked to read and sign a detailed consent form before proceeding (the study was approved by the HEC Montreal Research Ethics Board, no. 2018-2923).The EEG headset (Brainvision Acticap, 64 electrodes, 10-20 layout) was then installed on the participant’s head in accordance with the manufacturer’s instructions, which specified the choice of headset based on head size, proper positioning of the electrodes on the scalp, and gel application to lower impedance. Each electrode recorded signal at 250 Hz, and impedance was kept under 30 kOhm. Participants then sat comfortably ∼70 cm in front of a computer screen and keyboard.
Before the experiment began, participants were verbally instructed that they would have to choose from two drawings the one that represented a living being, using the left or right arrow on the keyboard. They were asked to answer as fast as possible while prioritizing accuracy over speed. The same instructions were also given on-screen before the practice round of eight stimuli to prevent misunderstanding. Identical instructions were given to all participants. After making sure the participant had no further questions concerning the procedure, we started the experiment.
Figure 1 illustrates the experimental procedure. Every stimulus was preceded by a neutral fixation cross, which was on screen for 0.5 seconds. A stimulus was then shown to the participant until one of the keyboard arrows was pressed, for a maximum duration of 2 seconds. If no response was given in the maximum time, the data were considered missing. All stimuli were presented twice to every participant for each block, and the experiment consisted of three blocks. In all cases, stimuli were always randomly presented. In total, each participant was asked to respond to 216 stimuli.
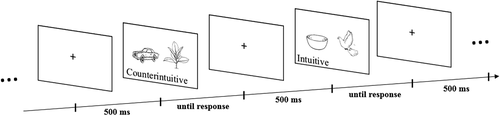
FIGURE 1. Timeline of the experimental procedure.
Stimuli
Stimuli consisted of pairs of line drawings, positioned at 2.5° to the right and 2.5° to the left of the center of the screen. The drawings could be of four categories: 1) living and moving things (fox, dove, or elephant); 2) living but nonmoving things (tree, flower, or plant); 3) nonliving and nonmoving things (bowl, lamp, or stool); and 4) nonliving but moving things (helicopter, car, or motorcycle). As shown in Figure 2, the pairs of drawings comprised both a living and nonliving object and both a moving and nonmoving object. The I stimuli consisted of pairs in which the living thing was moving. The CI stimuli consisted of pairs in which the living thing was not moving. The combination of drawings led to 9 I and 9 CI stimuli. Mirror stimuli were also created to control for the position of each image in each stimulus, leading to 18 I and 18 CI stimuli.
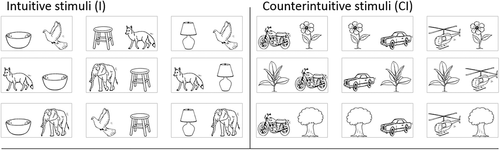
FIGURE 2. Combinations of images used for both I and CI stimuli.
Data Recording and Analysis
The stimuli were presented to the participants using E-prime 2.0 (Psychology Software Tools, Pittsburgh, PA). Behavioral data, such as accuracy and response times, were directly recorded via the same software. EEG data were collected using Brainvision Recorder software (Krigolson et al., 2017).
Behavioral data and EEG recordings were merged and processed using the Brainvision Analyzer 2 (Krigolson et al., 2017) software. A semiautomatic raw data inspection was run to remove artifacts from muscular sources. The data were then referenced using the Cz electrode, and a low-high cutoff filter of 0.5–60 Hz with a 60 Hz notch was applied. An independent component analysis was also run to remove eyeblinks and heartbeats. We segmented the data for all types of stimuli, using epochs of −100 to 500 milliseconds, before averaging all I and CI stimuli for each participant. Only the items that were answered successfully were kept for the analysis, as it is assumed that inhibitory control is the cognitive process that allows one to resist to the heuristic and give a scientific—hence successful—answer.
The signals from both I and CI conditions were statistically compared using the Brainstorm software (Tadel et al., 2011). We ran comparisons on all electrodes, as this study is to our knowledge the first one to investigate the implication of inhibitory control to overcome a life science heuristic using EEG. This bottom-up approach was chosen as a first step to get an overview of the entire signal distribution and to confirm the expected differences localizations between the conditions. All comparisons were made at a p < 0.01 threshold, with a Bonferroni correction for the number of electrodes. For this step, we only considered signal differences that lasted for 15 milliseconds. This choice was made in order to validate that the main differences in the signal were indeed localized only in the components of interest, and not on the whole signal.
As a second step, we ran top-down comparisons between conditions for the two components of interest. As mentioned previously, there does not seem to be an exact consensus regarding the N2 and LPP component latencies. Because the stimuli of this study were presented in a visual modality, it was expected that the N2 component would show around the 180-millisecond latency (Folstein and Van Petten, 2008). In line with the study of Colasante et al. (2017), we have chosen to select a time frame of 150–200 milliseconds for the N2 latency and 250–500 milliseconds for the LPP latency. The cognitive task used in the research of Colasante et al. (2017) has some similarities with the task of the present study, particularly in terms of the simplicity of the stimuli used. For this reason, we expected to observe the N2 and LPP components at a rather early latency (150–200 and 250–500 milliseconds, respectively). The literature also suggests that shorter latencies are often associated with superior mental performance in a task (Sur and Sinha, 2009). As our task implies identifying living things, which is a skill mastered at an early age, we assumed adults would perform very well, thus possibly showing early latency for both N2 and LPP components. For exhaustivity purposes, we ran the comparisons for each main scalp region, even though we did not have specific hypotheses targeting spatial localization. We used the average of all the electrodes located in a scalp region for both N2 and LPP components.
RESULTS
Behavioral Results
On average, participants were accurate in answering both I (M = 0.98, SD = 0.02) and CI (M = 0.91, SD = 0.03) stimuli. Response times were also rather short for correctly answered stimuli for both conditions (Mi = 464.73 milliseconds, SDi = 82.43 milliseconds; Mci = 495.71 milliseconds, SDci = 84.47 milliseconds). A correct answer consisted of identifying the living thing in each pair, while an incorrect answer consisted of selecting the nonliving thing or not responding for 2 seconds. Table 1 shows both accuracies and response times (RT) for all stimuli. A paired t test detected significant differences between I and CI stimuli both for accuracy, t(27) = 11.54, p < 0.001, and RT for accurately answered stimuli t(27) = 6.38, p < 0.001; CI>I. The difference between conditions had a very strong effect size for accuracy (d = 2.57) and a small to medium effect size for RT (d = 0.37).
| DV | Condition | N | M | SD | t | df | p | Cohen’s d |
|---|---|---|---|---|---|---|---|---|
| Accuracy | I | 28 | 0.98 | 0.02 | 11.54 | 27 | <0.001 | 2.57 |
| CI | 28 | 0.91 | 0.03 | |||||
| Response time | I | 28 | 464.73 | 82.43 | −6.38 | 27 | <0.001 | 0.37 |
| CI | 28 | 495.71 | 84.47 |
EEG Results
After removal of data contaminated by muscular artifacts, 97.1% of all successful stimuli were used in the analysis. More precisely, an average of 97.9 ± 2.2% of successful I stimuli and an average of 96.4 ± 3.3% of successful CI stimuli were included. We consider this level of inclusion very similar between our two conditions and of sufficient quality for the analysis.
Figure 3 illustrates the signal differences between both conditions between −100 and 500 milliseconds around stimuli presentation. Figure 3A shows the grand average waveforms, aggregated for all electrodes in each scalp region, and for both I and CI conditions. Figure 3B reports that 18 out of 64 electrodes showed significant differences between the conditions. Seventeen of these electrodes were located in or near the parietal region (P1, P2, P3, P4, P5, P6, P7, P8, Pz, CP2, CP3, CP4, CP6, PO7, PO8, TP7, TP8), three for the occipital region (O2, PO7, PO8), four for the frontal region (CP2, CP3, CP4, CP6), and two for the temporal region (TP7, TP8). Figure 3B also illustrates the significant t values between conditions for each electrode and the latencies for these significant differences. Figure 3C indicates that no other electrode showed significant differences between our conditions. For simplification purposes, we have chosen to represent on a single line all the results that were nonsignificant throughout the entire analyzed time frame, as these results were identical for all individual electrodes not mentioned (total of 46 electrodes).
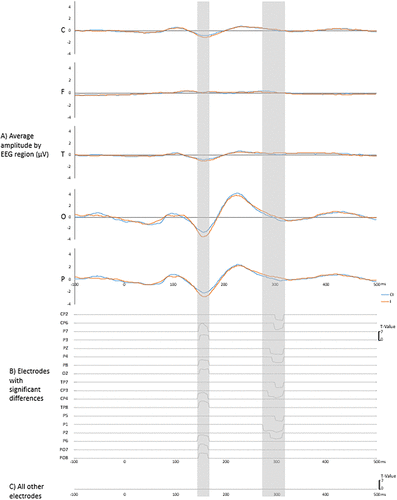
FIGURE 3. Signal differences between I and CI conditions. (A) Grand average of waveforms from all electrodes of each scalp region. (B) Significant statistical differences after stimuli exposure between conditions for each electrode with at least one significant difference. (C) Statistical differences after stimuli exposure between conditions for all other electrodes.
As highlighted in Figure 3, the significant differences between both conditions are only located around the N2/LPP components. The significant differences in the N2 negative peak were located between 142 and 166 milliseconds after stimulus presentation. The significant differences between conditions in the LPP region were located between 270 and 312 milliseconds after stimulus presentation. As can be seen in Figure 3B, the significant differences between I and CI conditions at the N2 peak were detected in nine electrodes; P3 (t = 4.29, p < 0.01), P6 (t = 5.34, p < 0.01), P7 (t = 6.85, p < 0.01), P8 (t = 4.60, p < 0.01), CP4 (t = 5.40, p < 0.01), TP8 (t = 5.32, p < 0.01), PO7 (t = 4.77, p < 0.01), PO8 (t = 4.20, p < 0.01), O2 (t = 4.07, p < 0.01). While those differences were almost all located in the close vicinity to the parietal region (eight electrodes out of nine), some of the significant differences were located at least partially in the occipital region (three electrodes), the central region (one electrode), and the temporal region (one electrode). As for the LPP component, the significant differences were detected in nine electrodes, also in the parietal region or very close by P1 (t = 6.33, p < 0.01), P2 (t = 4.89, p < 0.01), P4 (t = 4.66, p < 0.01), P5 (t = 4.10, p < 0.01), Pz (t = 4.80, p < 0.01), CP2 (t = 4.58, p < 0.01), CP3 (t = 5.85, p < 0.01), TP7 (t = 4.46, p < 0.01). While all significant electrodes were all at least partially located in the parietal region (nine electrodes), some of the significant differences were also located at least partially in the central region (three electrodes) and the temporal region (one electrode).
Figure 4 represents the statistical differences between I and CI conditions for both N2 and LPP components for each scalp region. The amplitude averages were calculated from the averaged signal of all significant and nonsignificant electrodes for each scalp region. As shown in Table 2, paired-sample t tests for the signal at the N2 latency reported higher amplitudes for I stimuli compared with CI stimuli in the parietal (t = 3.79, p < 0.01, d = 0.61), temporal (t = 2.99, p < 0.01, d = 0.43), occipital (t = 3.34, p < 0.01, d = 0.84), and central regions (t = 3.77, p < 0.01, d = 0.30). As for the LPP latency, a higher amplitude for I stimuli compared with CI stimuli was reported in the parietal region (t = 2.06, p < 0.05, d = 0.28).
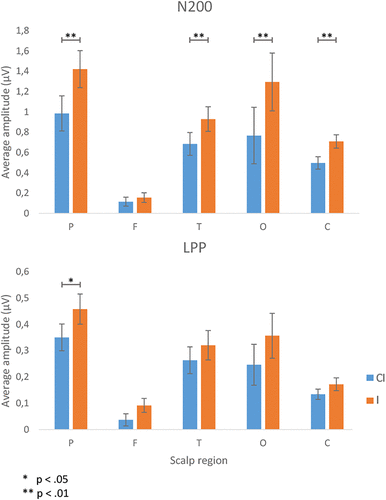
FIGURE 4. Average signal amplitudes of the N2 and LPP components for both conditions in each scalp region.
| Component | Region | Condition | N | M | SD | t | df | p | Cohen’s d |
|---|---|---|---|---|---|---|---|---|---|
| N2 | Parietal | C | 28 | 0.98 | 1.83 | 3.79 | 27 | 0.001 | 0.61 |
| I | 28 | 1.42 | 1.93 | ||||||
| Frontal | C | 28 | 0.12 | 0.46 | 1.03 | 27 | 0.312 | ||
| I | 28 | 0.16 | 0.50 | ||||||
| Temporal | C | 28 | 0.68 | 1.19 | 2.97 | 27 | 0.006 | 0.43 | |
| I | 28 | 0.93 | 1.29 | ||||||
| Occipital | C | 28 | 0.77 | 2.94 | 3.34 | 27 | 0.002 | 0.84 | |
| I | 28 | 1.29 | 3.02 | ||||||
| Central | C | 28 | 0.50 | 0.64 | 3.78 | 27 | 0.001 | 0.30 | |
| I | 28 | 0.71 | 0.69 | ||||||
| LPP | Parietal | C | 28 | 0.35 | 0.54 | 2.06 | 27 | 0.049 | 0.28 |
| I | 28 | 0.46 | 0.61 | ||||||
| Frontal | C | 28 | 0.04 | 0.24 | 1.74 | 27 | 0.094 | ||
| I | 28 | 0.09 | 0.28 | ||||||
| Temporal | C | 28 | 0.26 | 0.54 | 1.01 | 27 | 0.324 | ||
| I | 28 | 0.32 | 0.59 | ||||||
| Occipital | C | 28 | 0.25 | 0.82 | 1.49 | 27 | 0.148 | ||
| I | 28 | 0.36 | 0.90 | ||||||
| Central | C | 28 | 0.13 | 0.21 | 1.56 | 27 | 0.130 | ||
| I | 28 | 0.17 | 0.26 |
DISCUSSION
In this study, we aimed to observe the presence of inhibitory control in a life science task requiring the identification of moving or nonmoving living beings. More precisely, our hypothesis was that adults would show a typical EEG pattern of inhibitory control when successfully identifying things that are alive but not moving as living things as opposed to identifying things that are alive and moving. In summary, both behavioral and EEG data were analyzed and seem to confirm our hypothesis.
Persistence of the “Moving Things Are Alive” Heuristic into Adulthood
Behavioral Results.
The underlying hypothesis of this study was that simple heuristics such as “moving things are alive” may persist into adulthood and, in some cases, contribute to the persistence of misconceptions years after proper learning. These heuristics would thus need, in specific contexts, to be inhibited for the scientific conception to prevail. In our task, correctly identifying an object that is alive but not moving should thus require more time and cognitive work than properly identifying an object that is alive and moving. The level of reasoning complexity involved in a cognitive task is most often measured using accuracy rates and response times (Stavy and Babai, 2010; Zhu et al., 2019). Greater cognitive effort during a classification task would translate into two behavioral observations: 1) a lower accuracy rate in classifying CI stimuli due to the fact that they are not in line with the heuristic, thus more difficult; and 2) a longer response time for successfully classifying CI stimuli, because participants would have to suppress (inhibit) their intuitive conception to answer correctly.
Our behavioral results support those two predictions. Indeed, while accuracy rates in both CI and I conditions were high, we observed that the participants were significantly more accurate in the I condition (98%) than the CI condition (91%). The comparison of response times for successful trials further supports this result, as correctly identifying the living thing in the CI condition took significantly more time than doing so in the I condition, with a small to medium effect size (see Table 1). Those results thus indicate that adult participants answered less accurately and more slowly when presented with a stimulus that, as we hypothesized, would require them to resist the “moving things are alive” heuristic in order to answer according to the scientific conception. As mentioned previously, accuracy rates and response times are both considered to be indicators of cognitive effort and have been associated to inhibitory control in many specific contexts (Babai et al., 2010; Potvin et al., 2014, 2015; Potvin and Cyr, 2017). In the particular context of our study, we interpret these behavioral results as indicators of the persistence of the “moving things are alive” heuristic into adulthood and as an index of inhibitory control.
Furthermore, although response times are an indirect indicator of inhibition, these results are in line with complementary research in the field, which shows that the intuitive conception of living things based on objects’ mobility exists even when no time limit is imposed (Opfer and Siegler, 2004; Waxman, 2005). Our interpretation of those results thus aligns with existing literature (Babai and Amsterdamer, 2008; Shtulman and Valcarcel, 2012; Potvin et al., 2015), which supports the idea that students’ heuristics probably never really disappeared from their brains. They could remain encoded in neural circuits and coexist with scientific conceptions, despite their incompatibility in some contexts. According to this idea, some intuitive conceptions that are based on strong heuristics could persist even a very long time after learning the scientific concept. The “moving = alive” interference highlighted by our behavioral indicators (lower performance and longer response times) could be explained by the fact that, if the initial intuitive conception of living things really coexists with the scientific one, classifying a basic life science phenomenon associated with a strong heuristic will require a greater cognitive effort of inhibitory control in order to overcome the interference.
Hereby, our behavioral results support the idea of a coexistence between prior intuitive conception and scientific conceptions for the considered basic life science content matter and the need to exert inhibitory control to address the task scientifically. The results are also in line with other STEM conceptions tested using different methodologies, such as the concepts of buoyancy, electricity, or perimeter (Masson et al., 2012; Brault Foisy et al., 2015; Potvin et al., 2015; Babai et al., 2016; Potvin and Cyr, 2017).
EEG Results.
The EEG results are consistent with the behavioral results and also support the inhibition hypothesis. The ERP results indeed demonstrate significant differences between the conditions at two ERP components of interest for several scalp regions. First, the N2 component was larger for the CI stimuli (i.e., the stimuli requiring use of the scientific conception, in which the living thing is not moving). As mentioned in the Introduction, the N2 is considered in the literature to be an indicator of error detection, also referred to as conflict monitoring (Folstein and Van Petten, 2008; Dickter and Bartholow, 2010; Bockler et al., 2011; Liu et al., 2015). It is indeed a component that has been observed in traditional tasks used to measure inhibitory control such as flanker (Forster et al., 2011) and go-no-go (Folstein and van Petten, 2008) tasks: for instance, amplitudes of negativity at this component are larger for no-go trials that necessitate inhibitory control, than for go trials, particularly in the temporo-occipital region for visual stimuli (Folstein and van Petten, 2008).
Conflict monitoring is known to be involved as an initial step in inhibitory control (Botvinick et al., 2001; Botvinick, 2007; Waxer and Morton, 2011) that triggers processes of response inhibition and response selection. In the context of science education, previous studies that have investigated the role of inhibition using fMRI have observed a stronger involvement of the anterior cingulate cortex (ACC; see, e.g., Masson et al., 2012) in the evaluation of CI stimuli. The ACC is a brain area often associated with conflict detection (Bush et al., 1998; Botvinick et al., 2001, 2004; Botvinick, 2007; Menon et al., 2001; Monchi et al., 2001; Buchsbaum et al., 2005; Lie et al., 2006) that comes into play early in the inhibition process, allowing for the detection of conflict between two competing but coexisting representations (MacDonald et al., 2000; Garavan et al., 2002; Brault Foisy et al., 2015) and for the triggering of the inhibition response. The enhanced N2 component that we observe for CI stimuli is thus consistent with fMRI results of previous studies, as it has been suggested that the ACC activity is reflected in the N2 component, particularly in the central region (Van Veen and Carter, 2002). This suggests that the participants evaluated this type of stimuli as errors according to their common “moving things are alive” heuristic or that they evaluated it as something that conflicted with another conception, possibly the scientific one.
Although the N2 component appears early after the presentation of the stimulus in this study, this result is not uncommon. Indeed, the latency of N2 peak is known in certain cases to occur somewhere between 150 and 275 milliseconds following stimulus presentation (Nunez et al., 2017). In addition, age and cognitive abilities can change the timing of the N2 occurrence (Cintra et al., 2018). In the case of our study, the earliness of the N2 peak could be explained by the fact that the participants were adults and that the studied scientific conception was quite simple and had long since been acquired. It therefore appears unsurprising that the latency time between the presentation of the stimulus and the N2 is rather short.
Second, our results show that the LPP component was also greater for the evaluation of CI stimuli. According to several studies, the LPP is associated with conflict resolution and response selection (Coderre et al., 2011; Yin et al., 2016; Zhu et al., 2019). We therefore interpret this result as an indicator that, following the conflict detection (N2 component), the scientific knowledge was checked against the intuitive conception of living things and subsequently activated and selected so as to choose the right answer for the classification task.
In light of previous studies linking inhibitory control to scientific reasoning (Masson et al., 2012; Brault Foisy et al., 2015; Potvin et al., 2015; Babai et al., 2016; Potvin and Cyr, 2017), it appears reasonable to hypothesize that inhibitory control mechanisms might have played a role in the resolution of the conflict by suppressing the fast and intuitive response and allowing selection of the scientific one. This sequence of observations (LPP following N2) is also convergent with the EEG results obtained by Zhu and his team (2019) in the context of a task targeting a misconception about electricity. However, the LPP component is elicited here earlier than it was in Zhu et al.’s study (2019). This seems plausible, considering the early appearance of the N2 component: because conflict detection happened early, it seems logical to assume that conflict resolution also happened sooner.
Potential Implications for Life Science Education
Conceptual Change in Biology.
Our results point to the idea that the “moving things are alive” heuristic persists into adulthood. Two neural networks would continue to coexist in an adult who is well aware of what characterizes a living organism: a first neural network related to the intuitive conception of living things based on the “moving things are alive” heuristic and a second neural network related to the scientific conception that would have developed after learning. It is therefore likely that adults have not abandoned their initial based-on-heuristic intuitive conception but rather are able to actively suppress it to correctly complete the task pertaining to biology. Our interpretation is that adults, through their exposure to scientific knowledge of living organisms, have developed the ability to detect situations in which their heuristics interfere with the scientific concept, which, as has been shown for other conceptions, prompts the activation of the inhibition network (Potvin, 2013; Brault Foisy et al., 2015).
Results of the present study thus support a different perspective of cognitive and conceptual development than the one proposed by Piaget, according to which children’s development is incremental, with knowledge of increasing complexity being acquired through four discrete stages from childhood to adolescence (Piaget, 1954; Inhelder and Piaget, 1964). According to Piaget’s perspective, our results should have shown no difference in inhibition levels between the I and CI conditions, as the scientifically accurate knowledge would have overwritten the misconception. Our results instead suggest that several competing conceptions can coexist in the brain of an individual at any age (Shtulman and Valcarcel, 2012), hence the necessity to inhibit irrelevant conceptions in order to activate relevant ones (Siegler, 2007; Borst et al., 2015). Our results are thus more compatible with conceptual change models in which the learners’ initial conceptions (or pieces of such conceptions) remain accessible after undergoing a conceptual change (Mortimer, 1995; diSessa, 2006; Stavy et al., 2006; Bélanger, 2008; Ohlsson, 2009; Potvin, 2013) rather than models that imply that these intuitive conceptions are replaced by scientific ones (Nussbaum and Novick, 1982; Posner et al., 1982; Vosniadou, 1994). Indeed, if initial conceptions were eradicated or replaced during conceptual change, then we would neither have recorded longer response times for CI stimuli nor N2 and LPP ERP components. The detection of a conflict therefore seems to be an indicator of this interference caused by the heuristic. With regard to reasoning about living things, we provide here the first EEG evidence that the “moving things are alive” heuristic persists into adulthood, and we interpret our behavioral and EEG results as index of inhibitory control that may have allowed subjects to resist the heuristic while activating the correct scientific reasoning around the characteristics of living organisms.
In the context of science education, in this case biology, the concept of inhibitory control therefore sheds new light on and provides a more fundamental understanding of the students’ scientific learning pathways: instead of simply considering that students make mistakes, these mistakes can now be interpreted as the incorrect selection of an intuitive conception or as the use of an heuristic or automatism that they are unable to resist. According to this framework, children, adolescents, and adults can all make mistakes when they fail to inhibit the incorrect use of certain heuristics in specific contexts, which could explain why even experienced scientists can revert to naive reasoning when placed in a situation where they cannot exercise their inhibitory control (see, e.g., Kelemen et al., 2013).
In the case of the “moving things are alive” heuristic, despite seemingly trivial, it persists even after one has learned about living organisms, and it might still need to be inhibited in some contexts to allow for scientific reasoning. Although our research is unable to certainly determine the origins of this conceptual/cognitive difficulty, one avenue of explanation could be that the persistence of the intuitive conception might be due in part to the fact that its pragmatic truth is made relevant on a daily basis, as we humans are indeed alive and mobile and regularly interact with other human beings or animals that are also alive and mobile.
Pedagogical Implications.
We believe this study to be relevant to our more fundamental understanding of the acquisition processes for simple scientific concepts. On a practical level, it nevertheless presents pedagogical implications that are in line with recent literature on the prevalence of initial intuitive conceptions (Potvin et al., 2014; Brookman-Byrne et al., 2018). Given the alleged coexistence of contradictory heuristics or conceptions, it therefore appears logical to infer that putting energy into trying to eradicate or replace this intuitive misconception is somewhat useless. It would seem wiser to channel teaching efforts into helping the learners to reinforce their system 2 and learn to inhibit their system 1, if required. To do so, it appears relevant to help them slow down and develop and automatize the ability to recognize and inhibit their heuristic in various contexts. The same pedagogical logic would also apply to other intuitive conceptions based on similar heuristics.
Nevertheless, because inhibitory control is a central mechanism for overcoming several intuitive conceptions in science and other domains, it also appears important to determine how to promote the development and mobilization of inhibitory control in students, in addition to developing the understanding of the scientific conception. This would be the case in the specific context of life science education and, possibly, in science learning in general. In that sense, some research projects have already identified avenues of intervention that could facilitate the mobilization of inhibition (Houdé et al., 2011; Lubin et al., 2012; Rossi et al., 2012). The results of those research projects show that giving warnings to the students about the possible bias they need to overcome, combined with training to identify tempting but incorrect responses, would help students inhibit their spontaneous conceptions.
It should be noted that the detection and inhibition of heuristics and intuitive conceptions is only one tool among others to help students reason scientifically. Yet, in the context of students having trouble overcoming intuitive interference, the prevalence model of conceptual change developed by Potvin (2013; Potvin and Cyr, 2017) identifies operational propositions for achieving conceptual change in science that take into consideration previous studies on inhibitory control: 1) make the desired conception available at the very beginning of a teaching sequence, so that learners dispose of an additional response option; 2) introduce inhibitive warnings to make learners aware that their heuristics are insufficient in certain contexts and that they might be tempted to fall into a trap; and 3) make the prevalence of the desired conception last by repeatedly exercising the learners’ reflexes to inhibit a tempting but incorrect answer and by reinforcing and automatizing the desired scientific conception. Those propositions would benefit from being tested in multiple contexts of science education, including biology, and by contrasting them with other ways of seeing conceptual change.
CONCLUSIONS
In summary, we used response times and EEG to verify the hypothesis that the basic “moving things are alive” heuristic persists and might require inhibition in adults when evaluating CI stimuli as part of a classification task on living organisms in biology. The results of the present study are in line with previous response-time results indicating that children and adolescents (Babai et al., 2010), but also adults, must inhibit spontaneous heuristics like “moving things are alive” in order to correctly select which of two things is alive when one is not a moving thing. We indeed provide the first evidence that, for this type of stimuli, N2 and LPP ERP components are observed, which suggests that the “moving things are alive” heuristic still persist into adulthood and is inhibited in order to answer scientifically.
However, our scope being a specific intuitive conception of living things chosen for its known persistence in adolescence, it is not possible to generalize the results to all intuitive conceptions or to science learning in general. It is indeed plausible that the prevalence of different conceptions varies according to many individual factors such as age, development, or level of expertise or according to the characteristics of the misconceptions themselves (their origin, their strength, the frequency with which they are reinforced in everyday life, etc.). That being said, our results are concordant with many previous psychophysiological studies on various scientific misconceptions, such EEG results obtained by Zhu and colleagues (2019) and fMRI results (Masson et al., 2012) for the same misconception in electricity (“one wire is sufficient to light a bulb”) and for one in mechanics (“a heavier ball will fall faster than a lighter one”; Brault Foisy et al., 2015). They are also in agreement with several studies using response times to investigate misconceptions, for example, about buoyancy (“bigger objects sink more”; Potvin and Cyr, 2017; Skelling-Desmeules et al. 2018). It therefore seems that inhibitory control could be a key process in resisting several misconceptions at different ages. Future studies may seek to triangulate these results using EEG for other STEM conceptions in order to draw more solid conclusions, such as buoyancy (Potvin and Cyr, 2017)) or perimeter (Babai et al., 2016).
Another limitation of this study is that inhibitory control, like other cognitive processes, can only be inferred using indirect measures. The tools selected for this research are common and are validated in the scientific literature pertaining to inhibitory control in ERP contexts, particularly in terms of response times (Babai and Amsterdamer, 2008; Brookman-Byrne et al., 2018; Mason et al., 2019) but also EEG signals (Bockler et al., 2011; Zhu et al., 2013; Liu et al., 2015). Furthermore, this inability to directly measure inhibitory control can be mitigated if several recognized measures are triangulated and achieve similar results. Although this work focuses specifically on using EEG to identify the persistence of the heuristic and thus the need for inhibitory control, we have also used accuracy and response times and found that all results were consistent. This concordance among accuracy, response times, and EEG signals increases the reliability of our interpretation, thus reducing the impact of this limitation on our work.
We also acknowledge that our experimental design does not include any type of neutral stimuli, but only I and CI ones. Although neutral stimuli are often used for similar work in fields such as response-time studies, our design was deliberately limited to I and CI stimuli. This decision aimed to maximize the number of stimuli administered to participants under these two conditions considered to be most important, thereby increasing the statistical power of our study and the chances of detecting an effect on a heuristic acquired at a very early age and predicted to be hardly detectable in an adult population. Because the results suggest the persistence of the heuristic into adulthood, it would be appropriate to conduct the same experiment with other age groups, particularly young children, in whom we assume that the interference caused by the same heuristic would be stronger, this time adding a control condition.
Finally, this study was intended as an opportunity to use EEG to document the persistence of a heuristic as part of a cognitive task more similar to a school task. As mentioned earlier, the measurement of internal indirect constructs benefits greatly from the use of multiple tools to triangulate consistent results. The use of EEG as a tool to investigate the persistence of intuitive conceptions and the mobilization of inhibitory control mechanisms is an interesting asset for researchers in the field, as it offers a different range of qualities compared with fMRI, which has been used more frequently in the field until recently. In addition to the associated trade-off of exchanging excellent spatial resolution (fMRI) for excellent temporal resolution (EEG), the use of EEG provides an opportunity to administer conceptual tasks in environments closer to realistic science teaching contexts (Charland et al., 2015). This advantage is significant for science education in general, because conceptual change rarely occurs in a controlled context, such as a university laboratory, and would benefit from being studied in contexts as close as possible to authentic.
In addition, the use of portable EEG devices allows researchers to conduct their work directly in classrooms (Dikker et al., 2017; Xu and Zong, 2018). Several educational activities aimed at conceptual change could benefit from such a technology in order to better illuminate the persistence of different intuitive conceptions and the role of inhibitory control in the process, including computer simulations and educational video games (Skelling-Desmeules, 2018) or text reading (Xu and Zong, 2018). More generally, EEG could also investigate whether the order of activities in learning sequences can facilitate the use of inhibitory control by the student (Xu and Zong, 2018).
ACKNOWLEDGMENTS
This research project was supported by the Research Team in Science and Technology Education (EREST, UQAM) and by Tech3Lab (HEC), where the data collection took place. The authors declare that they have no potential conflict of interest in line with this project.